|
So, if 1000 crore atoms of hydrogen are arranged side by side, it will be 1 meter long. 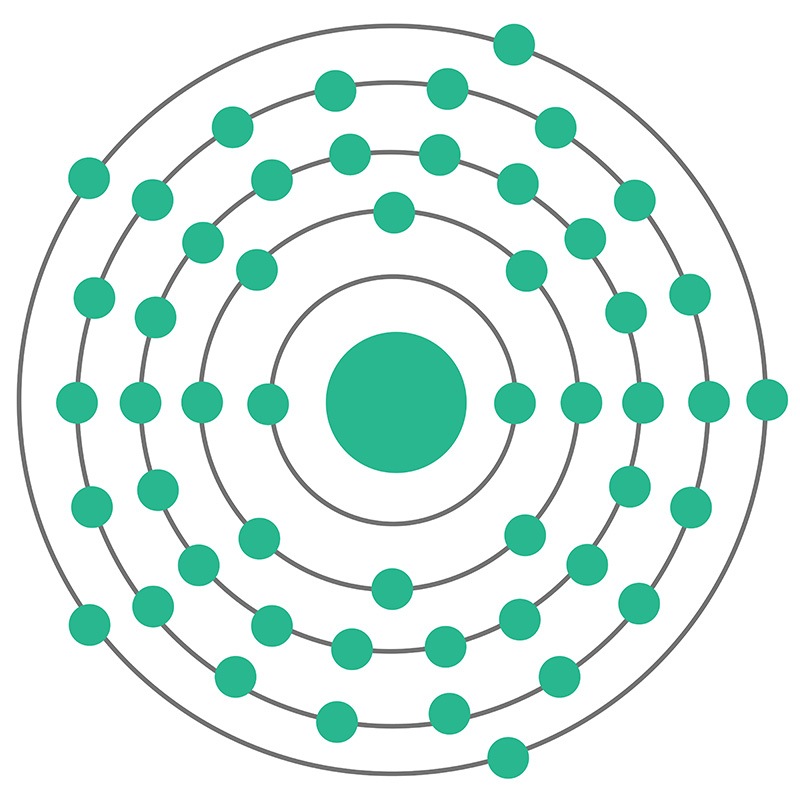
The diameter of an atom of hydrogen is 0.1nm (1.0nm = 10 -9m). Atoms are so small particles that they cannot be seen even under a powerful microscope. Where are the electrons, protons and neutrons located in an atom?Īn atom is the smallest particle of an element that has no independent existence but is directly involved in chemical reactions as the smallest unit. Hopefully, after reading this article you will know the details about this topic. This article discussed in detail how to easily find the number of protons, neutrons, and electrons in a polonium atom.Īlso discussed is the position of electrons, protons, and neutrons in an atom, the number of atomic masses, and the isotopes of polonium. The number of neutrons depends on the isotope of the element. 
Therefore, a polonium atom has one hundred twenty-five neutrons. The difference between the mass number of the polonium atom and the number of protons is one hundred twenty-five. The number of neutrons in an atom can be determined by the difference between the atomic mass and the number of protons. Therefore, a polonium atom has eighty-four protons and eighty-four electrons. The atomic number of an element is equal to the number of protons and electrons in that element. 
Polonium is the 84th element of the periodic table so its atomic number is 84. Left in direct contact with skin antimony can also cause irritation.Polonium is a classified metalloid and its symbol is ‘Po’. It can also cause irritation to the eyes. Breathing high levels of antimony for a long time can irritate the eyes and lungs, and can cause problems with the lungs, heart, and stomach, including vomiting, diarrheoa and stomach ulcers.Įxposure to high levels antimony from occurence in the workplace or contact with contaminated soils can cause difficulties with the heart, liver, lungs and stomach. How does antimony affect human health?Įxposure to antimony occurs in the workplace or from skin contact with soil at hazardous waste sites. Released to the atmosphere from natural sources and as a by-product of smelting lead and other metals, it generally drops to contaminate soils and waterways. 
HowĪlthough not abundant, antimony can be found in over 100 species of minerals. Small amounts are used in production of safety matches. The other major use is as antimony trioxide which is used for the production of flame retardant chemicals.Īntimony is used in the semi-conducter industry for certain silicone wafer, diode and infra-red detector productions. It is also used in automotive clutch and brake parts. MainĪntimony is used to increase the hardness of alloys, with lead alloys for batteries, with lead/copper/tin alloys for machine bearings. Number of Protons/Electrons in Antimony: 51Īntimony is a silvery-white brittle solid found in the earth's crust, estimates of the amount of antimony in the crust are between 0.2 and 0.5 ppm.Īlthough antimony resembles a metal it has poor electircal and conductive properties and does not react chemically like a metal and is classified as semi-metallic.Īntimony ores are mined and then mixed with other metals to form antimony alloys or combined with oxygen to form antimony oxide. anti plus monos - "a metal not found alone") antimony was first recognized in compounds and was recorded as a metal at the beginning of the 17th century and possibly prior to this date.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
